However, this does not explain the weak base ammonia (NH 3) which, in the presence of water, releases hydroxide ions into solution, but does not contain OH - itself. The theory suggests that in order for a substance to release either H or OH - ions, it must contain that particular ion. The Arrhenius theory has many more limitations than the other two theories. 
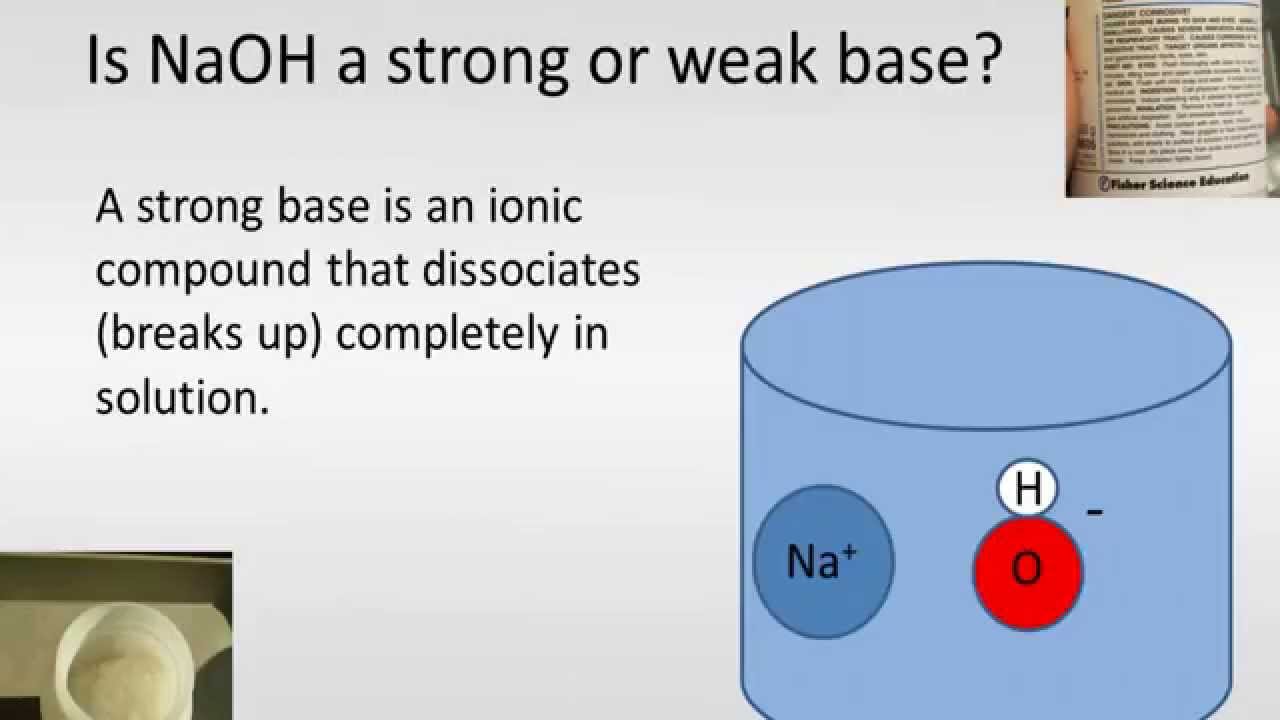
Thus, it is permissible to talk about “hydrogen ions” and use the formula H in writing chemical equations as long as you remember that they are not to be taken literally in the context of aqueous solutions. The equation "HA → H A –" is so much easier to write that chemists still use it to represent acid-base reactions in contexts in which the proton donor-acceptor mechanism does not need to be emphasized. The formula \(H_3O^ \) more adequately conveys the sense that it is both a molecule in its own right, and is also the conjugate acid of water. In a sense, \(H_2O\) is acting as a base here, and the product \(H_3O^ \) is the conjugate acid of water:Īlthough other kinds of dissolved ions have water molecules bound to them more or less tightly, the interaction between H and \(H_2O\) is so strong that writing “H (aq)” hardly does it justice, although it is formally correct. In the case of water, this will be the lone pair (unshared) electrons of the oxygen atom the tiny proton will be buried within the lone pair and will form a shared-electron (coordinate) bond with it, creating a hydronium ion, \(H_3O^ \). (Think of a pebble sitting in the middle of a sports stadium!) The resulting extraordinarily high charge density of the proton strongly attracts it to any part of a nearby atom or molecule in which there is an excess of negative charge. Although it carries only a single unit of positive charge, this charge is concentrated into a volume of space that is only about a hundred-millionth as large as the volume occupied by the smallest atom. The hydrogen ion in aqueous solution is no more than a proton, a bare nucleus. Owing to the overwhelming excess of \(H_2O\) molecules in aqueous solutions, a bare hydrogen ion has no chance of surviving in water. In this reaction, sodium hydroxide (NaOH) disassociates into sodium (Na ) and hydroxide (OH -) ions when dissolved in water, thereby releasing OH - ions into solution. The dissociation is represented by the following equation: The Brønsted-Lowry definition of acids and bases addresses this problem.Īn Arrhenius base is a compound that increases the concentration of OH - ions that are present when added to water. However, it does not explain why some substances that do not contain hydroxide ions, such as \(F^-\) and \(NO_2^-\), can make basic solutions in water. This theory successfully describes how acids and bases react with each other to make water and salts.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
